ORIGINAL ARTICLE
doi: 10.17533/udea.rccp.v29n2a06
Cryopreservation method and composition of the vitrification solution affect viability of in vitro bovine embryos¤
El método de criopreservación y composición de la solución de vitrificación afectan la viabilidad de embriones bovinos producidos in vitro
O método de criopreservação e a composição da solução de vitrificação afetar a viabilidade de embriões bovinos produzidos in vitro
José N Vargas Reyes*, MV, MSc; Liliana Chacón Jaramillo, MV, MSc, PhD.
Facultad de Ciencias Agropecuarias, Universidad de la Salle. Carrera. 7 No. 172-85. Bogotá, Colombia.
*Corresponding author: José N Vargas Reyes, Universidad de la Salle, Facultad de Ciencias Agropecuarias. Carrera 7 No 172-85, Bogotá, Colombia. E-mail: josvar8706@gmail.com
Received: November 13, 2014; accepted: January 27, 2016
Summary
Background: an optimal formulation for vitrifying in vitro-produced (IVP) bovine embryos is currently unavailable. Objective: to estimate whether differences in composition of vitrification solutions may affect the viability of IVP embryos as compared to that of embryos cryopreserved by a conventional slow-freezing method. Methods: bovine IVP embryos were cryopreserved by two methods: 1) a slow controlled-rate (1.5 M ethylene glycol (EG)), or 2) vitrification by using two different vitrification and thawing/warming solutions: (1) Protocol V1: commercial vitrification Kit, and (2) Protocol V2: defined vitrification (20% EG; 20% dimethyl sulfoxide (DMSO); 20% fetal bovine serum (FBS)) and warming (20% FBS; 0.2 M sucrose) solutions. Embryo viability was recorded at 24, 48, and 72 h after thawing/warming by evaluating the number of embryos that re-expanded and developed to the hatching blastocyst stage. Results: embryo survival rate was affected by the method of cryopreservation, where the frequency of embryos that re-expanded at 24 h after thawing/warming was higher for embryos vitrified with protocols V1 and V2 (89.0%, 86.2%, respectively) compared to those cryopreserved by the slow-controlled rate method (73.6%, p<0.05). Similarly, higher percentage of embryos cryopreserved by vitrification hatched at 72 h, where protocol V2 resulted in higher percentage of hatched embryos (84.3%) compared to protocol V1 (64,0%, p<0.05), and both were higher compared to the slowcontrolled rate method (55.2%, p<0.05). Conclusions: the method of cryopreservation and composition of the vitrification solution have a direct effect on the viability of bovine IVP embryos.
Keywords: ethylene glycol, expansion, hatching, slow freezing.
Resumen
Antecedentes: no existe una formula específica ideal para la congelación de embriones bovinos producidos in vitro (IVP). Objetivo: estimar si la composición de diferentes soluciones de vitrificación afecta la viabilidad de embriones bovinos IVP en comparación con embriones criopreservados mediante el método convencional de congelación lenta. Métodos: los embriones bovinos producidos in vitro se congelaron a través de los siguientes métodos: 1) congelación lenta (1,5 M de etilenglicol (EG)), 2) por vitrificación utilizando dos soluciones vitrificantes: (1) Protocolo V1: solución vitrificante comercial, (2) Protocolo V2: solución vitrificante (20% EG, 20% dimetil sulfóxido (DMSO), 20% suero fetal bovino (FBS)) y de calentamiento (20% FBS, 0,2 M sucrosa). La viabilidad embrionaria se evaluó a las 24, 48 y 72 h post-descongelación, a través de la medición del porcentaje de embriones que se expandieron y llegaron al estadio de eclosión. Resultados: el porcentaje de expansión embrionaria a 24 h fue más alto en los embriones que fueron vitrificados con los protocolos V1 y V2 (89,0%, 86,2%, respectivamente), que aquellos que fueron criopreservados con el método de congelación lenta (73,6%, p<0,05). Igualmente, un porcentaje mayor de embriones criopreservados por el método de vitrificación eclosionó a las 72 h, resultando en un mayor porcentaje en los embriones vitrificados con el protocolo V2 (84,3%), en comparación con el protocolo V1 (64,0%, p<0,05), y ambos porcentajes fueron más altos que los obtenidos con la congelación lenta (55,2%, p<0,05). Conclusiones: el método de congelación y la composición de la solución de vitrificación afectan la viabilidad post-congelación de los embriones bovinos IVP.
Palabras clave: congelación lenta, eclosión, etilenglicol, expansión.
Resumo
Antecedentes: não existe um meio criopreservante com uma composição perfeita para a vitrificação de embriões bovinos in vitro (PIV). Objetivo: avaliar se a composição de diferentes soluções de vitrificação afeta a viabilidade de embriões bovinos PIV, em comparação com embriões criopreservados pelo método convencional de congelação lenta. Métodos: embriões bovinos PIV foram criopreservados por dois métodos: 1) congelação lenta controlada (1,5 M etilenoglicol (EG)), 2) vitrificação com duas soluções diferentes: (1) Protocolo V1: Kit comercial de vitrificação, (2) Protocolo 2: soluções definidas de vitrificação (20% EG; 20% dimetilsulfóxido (DMSO); 20% soro fetal de bovino (FBS)) e aquecimento (20% FBS; 0,2 M sacarose). A viabilidade do embrião foi avaliada pelo número de embriões re-expandidos e desenvolvidos até o estágio de blastocisto expandido após 24, 48 e 72 h descongelamento/aquecimento. Resultados: a taxa de sobrevivência dos embriões afetou-se pelo método de criopreservação: a frequência dos embriões re-expandidos após 24 h de aquecimento/descongelamento foi maior nos embriões vitrificados com os protocolos V1 e V2 (89% e 86,2%; respectivamente), quando foi comparada com a frequência dos embriões criopreservados pelo método de congelação lenta (73,6%, p<0,05). Uma maior porcentagem de embriões criopreservados por vitrificação eclodiu após 72 h. O protocolo V2 apresentou maior porcentagem de embriões eclodidos (84,3%) quando comparado com o protocolo V1 (64%, p<0,05), ambos apresentaram maior porcentagem de eclosão quando comparados ao método de congelação lenta (55,2%; p<0,05). Conclusões: o método de criopreservação e composição da solução de vitrificação têm um impacto direto na viabilidade dos embriões bovinos PIV.
Palavras chave: congelamento lento, eclosão, etilenoglicol, expansão.
Introduction
Cryopreservation of in vitro-produced (IVP) embryos plays an important role protecting genetic material in traditional cattle breeding systems (Ha et al., 2014). Approximately 9.5% of the 385,000 IVP bovine embryos transferred worldwide in 2012 were cryopreserved (Perry, 2012). The low number of cryopreserved embryos could be explained by the fact that IVP embryos are, to some extent, developmentally compromised as compared to their in vivo-derived (IVD) counterparts. As IVP embryos have poor survival rates after cryopreservation (Rizos et al., 2003), they are associated with reduced pregnancy rates (Hasler, 2003). Hence, if such embryos are to be used for commercial bovine reproduction, cryopreservation techniques must be improved (Xu et al., 2006).
During cryopreservation, cells are suspended in a suitable solution, then cooled, stored in liquid nitrogen, warmed to room temperature, and returned to a physiological solution. During each step of this process, cells are at risk of various injuries, such as formation of intracellular ice during cooling (Pollard and Leibo, 1994) which affects the relative abundance of developmentally important gene transcripts (Wrenzycki et al., 2007) and fetal viability after embryo transfer (ET; Schmidt et al., 1996).
Slow-rate freezing and vitrification have been used to cryopreserve IVP bovine embryos. The principles and methodologies for slow-rate freezing and vitrification have been described elsewhere (Saragusty and Arav, 2011; Vajta et al., 2013). The slow freezing method has been widely used for cryopreserving IVP and IVD embryos; however, this method requires special equipment and a longer freezing time (Naik et al., 2005). In contrast, vitrification does not require expensive or sophisticated equipment, but it does require technical expertise to do it properly (Vajta and Nagy, 2006).
In terms of embryo survival after thawing/ warming, vitrification of IVP embryos has been proved to be at least as efficient (Inaba et al., 2011) or better (Vajta and Nagy, 2006; Trigal et al., 2012; Vajta, 2013) than slow-rate freezing, so it is not easy to decide for one methodology over the other; each laboratory should decide which one adapts better to their conditions (Caamaño et al., 2015).
The objective of the present study was to estimate whether differences in composition of vitrification solutions affect in vitro viability of bovine IVP embryos as compared to that of embryos cryopreserved using a conventional slow-freezing method.
Material and methods
Ethical considerations
The study followed the guidelines for animal care and use of Universidad de la Salle, Colombia (Bogotá; Protocol 002, from March 13, 2012).
Chemicals
All chemicals were provided by Sigma-Aldrich (St. Louis, MO, USA) unless otherwise stated.
In vitro production (IVP) of bovine embryos
In vitro oocyte maturation (IVM). Bovine ovaries were obtained from a local abattoir within 15 min post-slaughter and transported to the laboratory at 36-38 °C within 3-4 h in saline solution (0.9% NaCl). The ovaries were rinsed twice in phosphate-buffered saline (PBS) and follicular fluid aspirated from 3 to 6 mm follicles using a 21 gauge needle and a 10 mL syringe. Cumulus oocyte complexes (COCs) were collected from the follicular fluid and those with three or more layers of cumulus cells and homogeneous cytoplasm were randomly distributed in groups of 20-25 COCs and cultured in 100 μL droplets of IVMmedium. IVM consisted of tissue culture medium 199 (TCM-199) supplemented with 2.5 mM Na-pyruvate, 100 IU/mL penicillin, 100 μg/mL streptomycin, 1 mg/ mL estradiol -17 β, 0.01 IU/mL follicle stimulating hormone (FSH), 0.1 IU/mL luteinizing hormone (LH), and 10% fetal bovine serum (FBS) for 24 h in a humidified atmosphere of 5% CO2 in air at 39 °C.
In vitro fertilization (IVF). Frozen-thawed semen from a Holstein bull of proven fertility was used for IVF. Straws were thawed in a water bath at 37 °C for 30 sec and sperm suspension layered onto a two-layer (45:90%) discontinuous Percoll gradient solution (Amersham, Pharmacia Biotech AB, Sweden) by centrifugation (500 x g) for 10 min. The supernatant was removed and the sperm pellet was re-suspended in Tyrode’s Albumin-Lactate-Pyruvate (TALP) IVF medium. For IVF, motile spermatozoa (~1 x 106 /mL) were co-incubated with COCs (6 to 7) in 5% CO2 in air at 39 °C under mineral oil in 25 μL droplets of TALP- IVF medium for 18 h.
In vitro culture (IVC). After IVF, the remaining cumulus and corona cells were removed by gently vortexing. Then, presumptive zygotes were cultured in 25 μl droplets (1 embryo/μL) of synthetic oviductal fluid medium (SOF) as previously described by Holm et al. (1999), covered with mineral oil in a humidified atmosphere (5% CO2, 5% O2 and 90% N2) at 38.5 °C.
Embryo cryopreservation
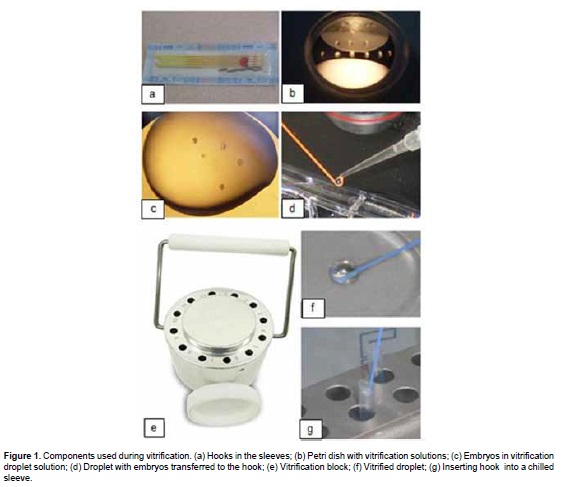
On day 6 or 7 of IVC, embryos at the morula, early blastocyst and blastocyst stage were cryopreserved either: 1) at a slow controlled-rate (control group), or 2) through vitrification by using two different vitrification and warming solutions: (1) Protocol V1: a commercial vitrification Kit from Achilles Genetics® (of unknown composition), and (2) Protocol V2: vitrification and warming solutions that were prepared in the laboratory. Embryos in both vitrification protocols were cryopreserved by using the vitrification hook WTA system (WTA, Watanabe Applied Technology, WTA, Brazil). The system consisted of a hook device and three pieces of equipment: 1) foam bath, 2) plastic container placed inside the foam bath, and 3) vitrification block placed inside the plastic container for cooling the embryos. The vitrification block was cooled by filling the foam bath with liquid nitrogen to maintain the temperature during the vitrification process.
Vitrification - Protocol V1
Vitrification and warming solutions were carried out in 5 μL micro-drops of each in Petri dishes at 22 °C. One to four embryos were exposed to an equilibration solution for 2 min, and then transferred into a vitrification solution (VS1) for 2 min, to be finally transferred into VS2 for 45 s. Embryos were loaded onto the hook device which was then held against the cooled surface of the vitrification block until the droplet vitrifies into a glassy bead. The hook was placed immediately into pre-cooled sleeves and plunged into liquid nitrogen (components of the system are shown in Figure 1).
After storage, embryos were warmed by immersion of the hook device directly into warming solution (WS1) for 2 min, which was followed by transferring them into a WS2 for 2 min (Figure 2).
Warmed embryos were cultured in 50 μl drops of SOF medium under paraffin oil (5-7 embryos/droplet) at 39 °C in a humidified atmosphere of 7% O2, 5% CO2, and 90% N2. Embryo viability was recorded at 24, 48, and 72 h after thawing by evaluating the numbers of embryos that re-expanded to their original size and developed to the hatching blastocyst stage.
Vitrification - Protocol V2
Embryo handling was performed at 22 °C while embryo culture media was maintained at 39 °C. The vitrification solution (VS2) consisted of 20% (v/v) ethylene glycol (EG; E9129), 20% (v/v) dimethyl sulfoxide (DMSO; D5879), and 20% (v/v) FBS diluted in TCM-199. One to four embryos were equilibrated in TCM- 199 supplemented with 20% FBS for 5 min. Then, embryos were transferred to VS1 consisting of TCM-199 + 20% FBS, + 10% EG, and 10% DMSO for 3 min, and finally briefly exposed (45 s) to the VS2. Embryo loading and cooling was performed as described in Protocol V1. For warming, embryos were transferred directly into 500 μL TCM- 199 supplemented with 20% FBS and 0.2 M Sucrose (S; S9378). Embryo culture and viability evaluation were performed as described in Protocol V1.
Slow controlled-rate freezing
Embryos were frozen according to a method previously reported by Lim et al. (2008). Briefly, embryos at each developmental stage were equilibrated in a cryoprotectant solution consisting of 1.5 M EG, 0.1 M S and 10% FBS in D-PBS for 5 min at room temperature. Embryos were individually loaded into 0.25 mL straws and, after sealing the tip, each straw was placed into the chamber of a controlled rate freezing unit (model CL-5500, CryoLogic, Victoria, Australia) equilibrated for 10 min at -7 °C during which time seeding was initiated. Freezing was accomplished at a cooling rate of 0.3 °C/min from -7 to -35 °C, and straws were plunged into liquid nitrogen for storage.
Straws were thawed for 6 to 10 s in air at 20 °C, followed by 15 s in a water bath at 37 °C. Thawed embryos were washed three times for 4 min each in Hepes buffered TCM-199 and then cultured in SOF medium. Embryo viability was recorded at 24, 48, and 72 h after thawing by evaluating the number of embryos that developed to the expanded or hatching blastocyst stage.
Experimental designs
Morulae, early blastocysts and in vitro produced blastocysts were randomly allocated to three freezing protocols: slow controlled-rate freezing, vitrification protocol V1, and vitrification protocol V2. Embryo survival was evaluated in terms of re-expansion and hatching at 24, 48, and 72 hours after thawing/warming.
Statistical analysis
Nonparametric Chi Square Test was used for reexpanded and hatching rate using the STATISTICS 8.0. program (Analytical Software in Tallahasee, Florida). For all analysis, p<0.05 was considered significant.
Results
The frequency of embryos that re-expanded at 24 h after warming/thawing was higher for embryos vitrified with protocols V1 and V2 (89.0, 86.2%, respectively), compared with those cryopreserved by slow-controlled rate method (73.6%, p<0.05; Table 1). Similarly, higher percentages of embryos cryopreserved by vitrification hatched at 72 h after warming/thawing, where protocol V2 resulted in higher percentage of hatched embryos (84.3%) compared with protocol V1 (64.0%, p<0.05). Furthermore, hatching rates in both vitrification solutions were higher compared with embryos cryopreserved by the slow-controlled rate method (55.2%; p<0.05; Table 1).
Although re-expansion rates observed after 24 h of warming for embryos vitrified with V1 and V2 protocols were higher than those of embryos in the slow-controlled rate group, re-expansion rates at 72 h for embryos vitrified with protocol V1 (31.2%) were similar to those of embryos cryopreserved by slowcontrolled rate (21.9%), but re-expansion rate in both groups was higher than that of embryos vitrified with protocol V2 (9.8%; p<0.05; Figure 3). In contrast, there were no significant differences in re-expansion rates among treatments at 48 h (Figure 3).
The percentage of vitrified embryos that hatched at 24 h after warming was similar for vitrification protocols V1 (48.4%) and V2 (36.2%), and higher than that of embryos frozen by the slow-controlled rate method (21.5%; p<0.05; Figure 4). The percentages of blastocysts that hatched at 48 h after warming was similar for embryos vitrified with protocol V2 and frozen by slow freezing (41.1 and 30.7%, respectively), but higher than those vitrified with protocol V1 (9.5%; p<0.05; Figure 4). The lowest hatching rates were observed at 72 h after warming/ thawing and no differences were observed among treatments (Figure 4).
Discussion
In the present study we evaluated the effect of slow freezing and vitrification techniques on the survival of in vitro bovine embryos produced under the same culture conditions. Results show that there are morphologic differences regarding re-expansion and hatching rates. We observed that bovine IVP embryos survived vitrification at higher rates than freezing by the slow-controlled rate method. Previous studies have shown similar results (Nedembale et al., 2004; Kuwayama , 2007; Rios et al., 2010; Trigal et al., 2012; Zhao et al., 2012), and demonstrated that vitrification is an appropriate method for cryopreservation of bovine IVP embryos (Vajta, 2000).
Additionally, vitrification with a V2 solution prepared at our laboratory resulted in higher percentage of embryos reaching the hatching stage compared with embryos vitrified with the commercially available V1 solution. We infer that the differences could be linked directly with the development stage of the embryo, volume and concentration of the cryoprotectants. It seems that these factors have a severe impact on embryo cryopreservation (Rios et al., 2010; Shirazi et al., 2010; Stinshoff et al., 2011).
On the other hand, it is not clear why embryos cryopreserved by vitrification resulted in higher survival rates than that of slow-controlled rate freezing. It is possible that the higher embryo survival rates at 24 h post-warming for embryos vitrified in both vitrification solutions (V1 or V2) resulted from inhibition of chilling injury by the ultra-rapid cooling rates and lower cellular damage during cryopreservation (Vajta et al., 1998).
Despite vitrification avoids problems associated with ice crystal formation, this method implies a challenge derived from the high concentration of cryoprotectant. However, the observed low proportion of cells with altered plasma membrane and their relation with expansion and hatching rates may be attributed to the brief exposure to cryoprotectant, as described by Vajta et al. (1998) and Mucci et al. (2006). Membrane stability of in vitro produced bovine embryos might be an important factor determining their susceptibility and viability to freezing (Imai et al., 1997).
A possible adverse effect of cryopreservation is the reduced ability of post-warming embryos to continue mitotic division (Kaidi et al., 1998), or the induction of apoptosis or necrosis in some embryonic cells (Abdalla et al., 2010). The adverse effect of vitrification/warming in this study was consistent with the commonly reported losses of these processes, which is in agreement with results reported for cows (George et al., 2006; Abdalla et al., 2010; Ha et al., 2014).
In conclusion, the method of cryopreservation and the composition of the vitrification solution have a direct effect on the viability of bovine IVP embryos. Results of this work, however, require verification by embryo transfer.
Acknowledgements
This study was supported by Universidad de la Salle and Vitrogen Laboratory– Bogotá, Colombia.
Conflicts of interest
The authors declare they have no conflicts of interest with regard to the work presented in this report.
Notes
¤To cite this article: Vargas JN, Chacón L. Cryopreservation method and composition of the vitrification solution affect viability of in vitro bovine embryos. Rev Colomb Cienc Pecu 2016; 29:130-137.
References
Abdalla H, Shimoda M, Hara H, Morita H, Kuwayama M, Hirabayashi M, Hochi S: Vitrification of ICSI-and IVF-derived bovine blastocysts by minimum volume cooling procedure: effect of developmental stage and age. Theriogenology 2010; 74:1028-1035.
Caamaño J, Gómez E, Trigal B, Muñoz M, Carrocera S, Martín D, Díez C. Survival of vitrified in vitro–produced bovine embryos after a one-step warming in-straw cryoprotectant dilution procedure. Theriogenology 2015; 83(5):881-890.
George F, Vrancken M, Verhaeghe B, Verhoeye F, Schneider Y-J, Massip A, Donnay I. Freezing of in vitro produced bovine embryos in animal protein-free medium containing vegetal peptones. Theriogenology 2006; 66:1381-1390.
Ha AN, Park HS, Jin JI, Lee SH, Ko DH, Lee DS, Kong IK. Postthaw survival of in vitro-produced bovine blastocysts loaded onto the inner surface of a plastic vitrification straw. Theriogenology 2014; 81:467-473.
Hasler JF. The current status and future of commercial embryo transfer in cattle. Anim Reprod Scie 2003; 79:245-264.
Holm P, Booth PJ, Schmidt MH, Greve T, Callesen H. High bovine blastocyst development in a static in vitro production system using SOFaa medium supplemented with sodium citrate and myo-inositol with or without serum-proteins. Theriogenology 1999; 52:683-700.
Imai K, Kobayashi S, Goto Y, Dochi O, Shimohira I. Cryopreservation of bovine embryos obtained by in-vitro culture of IVM-IVF oocytes in the presence of linoleic acid albumin. Theriogenology 1997; 47:347-347.
Inaba Y, Aikawa Y, Hirai T, Hashiyada Y, Yamanouchi T, Misumi K, Ohtake M, Somfai T, Kobayashi S, Saito N, Matoba S, konishi K, Imai K. In-straw cryoprotectant dilution for bovine embryos vitrified using cryotop. J Reprod Dev 2011; 57:437-443.
Kaidi S, Donnay I, Van Langendonckt A, Dessy F, Massip A. Comparison of two co-culture systems to assess the survival of in vitro produced bovine blastocysts after vitrification. Anim Reprod Sci 1998; 52:39-50.
Kuwayama M. Highly efficient vitrification for cryopreservation of human oocytes and embryos: the Cryotop method. Theriogenology 2007; 67:73-80.
Lim KT, Jang G, Ko K H, Lee WW, Park HJ, Kim JJ, Lee BC. Improved cryopreservation of bovine preimplantation embryos cultured in chemically defined medium. Anim Reprod Sci 2008; 103:239-248.
Mucci N, Aller J, Kaiser GG, Hozbor F, Cabodevila J, Alberio RH. Effect of estrous cow serum during bovine embryo culture on blastocyst development and cryotolerance after slow freezing or vitrification. Theriogenology 2006; 65:1551-1562.
Naik BR, Rao BS, Vagdevi R, Gnanprakash M, Amarnath D, Rao V. Conventional slow freezing, vitrification and open pulled straw (OPS) vitrification of rabbit embryos. Anim Reprod Sci 2005; 86:329-338.
Nedembale TL, Dinnyés A, Groen W, Dobrinsky J R, Tian XC, Yang X. Comparison on in vitro fertilized bovine embryos cultured in KSOM or SOF and cryopreserved by slow freezing or vitrification. Theriogenology 2004; 62:437-449.
Perry G. 2012 statistics of embryo collection and transfer in domestic farm animals. Data retrieval Committee reports, International Embryo transfer Society, IETS December 2013.
Pollard JW, Leibo SP. Chilling sensitivity of mammalian embryos. Theriogenology 1994; 41:101-106.
Rios GL, Mucci NC, Kaiser GG, Alberio RH. Effect of container, vitrification volume and warming solution on cryosurvival of in vitro-produced bovine embryos. Anim Reprod Sci 2010; 118:19-24.
Rizos D, Gutierrez-Adan A, Perez-Garnelo S, De La Fuente J, Boland MP, Lonergan P. Bovine embryo culture in the presence or absence of serum: implications for blastocyst development, cryotolerance, and messenger RNA expression. Biol Reprod 2003; 68:236-243.
Saragusty J, Arav A. Current progress in oocyte and embryo cryopreservation by slow freezing and vitrification. Reproduction 2011, 141(1):1-19.
Schmidt M, Greve T, Avery B, Beckers JF, Sulon J, and Hansen HB. Pregnancies, calves and calf viability after transfer of in vitro produced bovine embryos. Theriogenology 1996; 46:527-539.
Shirazi A, Soleimani M, Karimi M, Nazari H, Ahmadi E, Heidari B: Vitrification of in vitro produced ovine embryos at various developmental stages using two methods. Cryobiology 2010, 60:204-210.
Statistix 8, 2003. Statistix8: Analytical software user’s manual. Tallahassee, Florida.
Stinshoff H, Wilkening S, Hanstedt A, Brüning K, Wrenzycki C: Cryopreservation affects the quality of in vitro produced bovine embryos at the molecular level. Theriogenology 2011, 76:1433-1441.
Trigal B, Gómez E, Caamaño JN, Muñoz M, Moreno J, Carrocera S, Martin D, Diez C. in vitro and in vivo quality of bovine embryos in vitro produced with sex-sorted sperm. Theriogenology 2012; 78:1465-1475.
Vajta G. Holm P, Kuwayama M, Booth PJ, Jacobsen H, Greve T, Callesen H. Open pulled straw (OPS) vitrification: a new way to reduce cryoinjuries of bovine ova and embryos. Mol Reprod Dev 1998; 5:53-58.
Vajta G. Vitrification of the oocytes and embryos of domestic animals. Anim Reprod Sci 2000; 60:357-364.
Vajta G, Nagy ZP. Are programmable freezers still needed in the embryo laboratory? Review on vitrification. Reprod BioMed Online 2006; 12:779-796.
Vajta G. Vitrification in human and domestic animal embryology: work in progress. Reprod Fert Dev 2013; 25:719-727.
Wrenzycki C, Herrmann D, Niemann H. Messenger RNA in oocytes and embryos in relation to embryo viability. Theriogenology 2007; 68:S77-S83.
Xu J, Guo Z, Su L, Nedambale T, Zhang J, Schenk J, Moreno J, Dinnyés A, Ji W, Tian X. Developmental potential of vitrified Holstein cattle embryos fertilized in vitro with sex-sorted sperm. J Dairy Sci 2006; 89:2510-2518.
Zhao Xm, Du Wh, Wang D, Hao Hs, Qin T, Liu Y, Zhu Hb. Controlled freezing and open-pulled straw (OPS) Vitrification of in vitro produced bovine blastocysts following analysis of ATP content and reactive oxygen species (ROS) level. J Integr Agr 2012; 11:446-455.