Enfermedades virales transmitidas por garrapatas
DOI:
https://doi.org/10.17533/udea.iatreia.v31n1a04Palabras clave:
arbovirus, enfermedades transmitidas por garrapatas, Latinoamérica, salud pública, zoonosisResumen
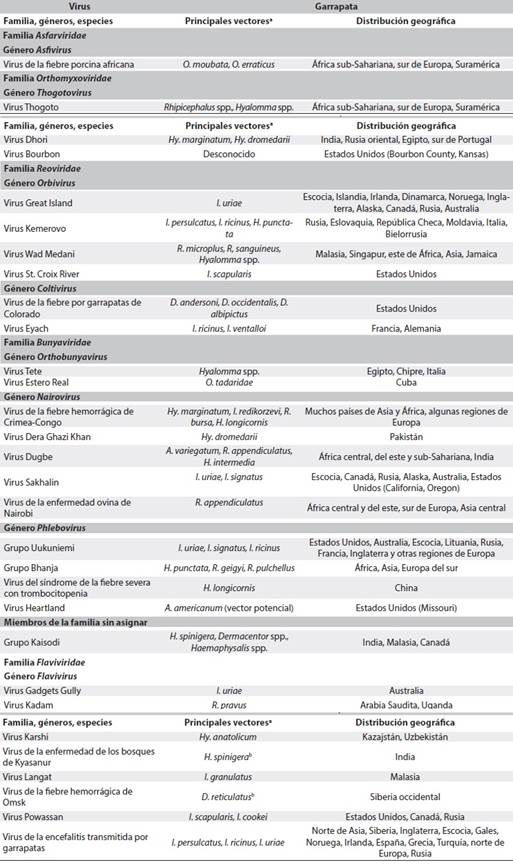
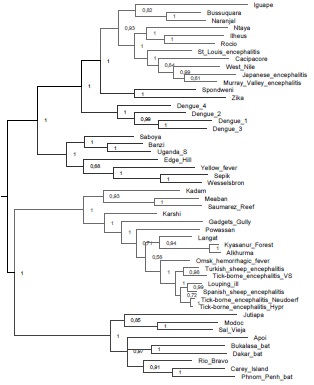
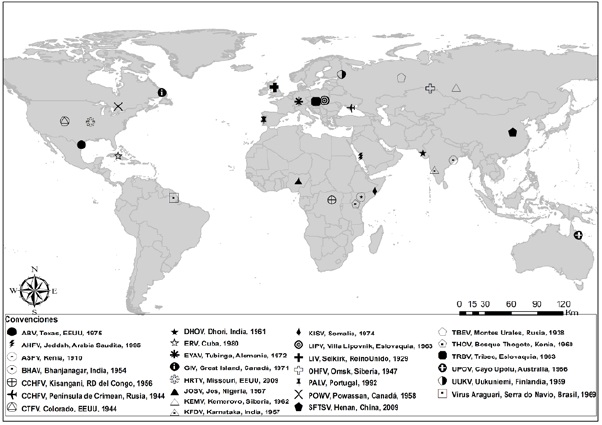
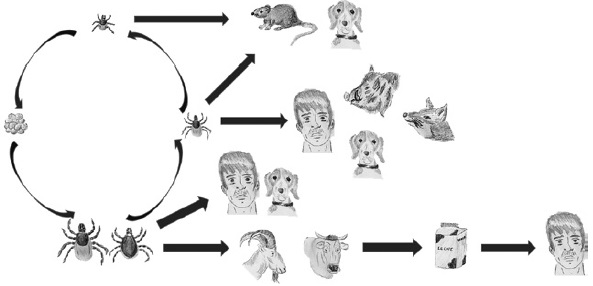
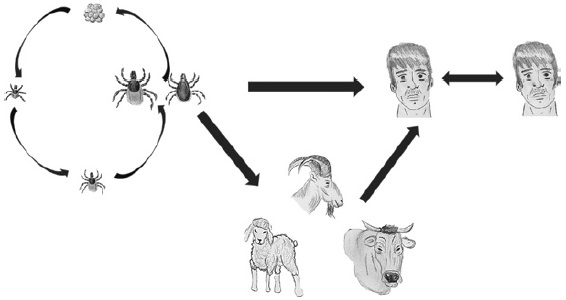
Los virus transmitidos por garrapatas (VTG) pertenecen a las familias Flaviviridae, Bunyaviridae, Reoviridae, Asfarviridae y Orthomyxoviridae y son agentes causales de diferentes enfermedades en humanos y animales. Debido a la creciente importancia epidemiológica que están teniendo los VTG, esta revisión pretende englobar el conocimiento actual de estos agentes y las enfermedades que producen, así como exponer las estrategias abordadas en prevención y tratamiento que se han implementado hasta el momento en diferentes países. Es evidente que para la región Neotropical hacen falta estudios sobre los VTG presentes en la región, ya que la gran mayoría de los artículos, tanto revisiones de tema como trabajos originales, presentan datos de las regiones Neártica y Paleártica. Considerando el panorama actual de los estudios de VTG en la región Neotropical y las particularidades de la misma, es muy probable que existan otros VTG aún no identificados que podrían tener algún impacto en salud pública.
Descargas
Citas
(1.) Labuda M, Nuttall P. Viruses transmitted by ticks. In: Bowman A, Nuttall P, editores. Ticks: biology, disease and control. United Kingdom: Cambridge University Press; 2008, p. 253-80.
(2.) World Health Organization. Vaccines against tickborne encephalitis: WHO position paper? Weekly epidemiological record [Internet] 2011[cited 2016 Nov 30];86(24): [241-56]. Available from: http://www.who.int/wer/2011/wer8624.pdf
(3.) Organización Mundial de la Salud [Internet]. Ginebra, Suiza: OMS; 2013 [consultado 2016 Nov 30]. Centro de prensa: Fiebre hemorrágica de Crimea-Congo. Disponible en: http://www.who.int/mediacentre/factsheets/fs208/es/
(4.) Konstantinovich D, Yurievich M, Vladimirovich S, Grigorievich P. Zoonotic viruses of northern Eurasia: Taxonomy and Ecology. Moscú: Elsevier Science; 2015.
(5.) Organización Mundial de la Salud [Internet]. Ginebra, Suiza: OMS; 2016. Centro de prensa: Enfermedades transmitidas por vectores [Internet]. [consultado 2016 Nov 30]. Disponible en: http://www.who.int/mediacentre/factsheets/fs387/es/
(6.) Estrada-Peña A, Ayllón N, de la Fuente J. Impact of climate trends on tick-borne pathogen transmission. Front Physiol. 2012 Mar;3:64. DOI 10.3389/fphys.2012.00064.
(7.) Dupuis AP 2nd, Peters RJ, Prusinski MA, Falco RC, Ostfeld RS, Kramer LD. Isolation of deer tick virus (Powassan virus, lineage II) from Ixodes scapularis and detection of antibody in vertebrate hosts sampled in the Hudson Valley, New York State. Parasit Vectors. 2013 Jul;6:185. DOI 10.1186/1756-3305-6-185.
(8.) Johnson AJ, Karabatsos N, Lanciotti RS. Detection of Colorado tick fever virus by using reverse transcriptase PCR and application of the technique in laboratory diagnosis. J Clin Microbiol. 1997 May;35(5):1203-8. Erratum in: J Clin Microbiol 1997 Jul;35(7):1920.
(9.) Matsuno K, Weisend C, Kajihara M, Matysiak C, Williamson BN, Simuunza M, et al. Comprehensive molecular detection of tick-borne phleboviruses leads to the retrospective identification of taxonomically unassigned bunyaviruses and the discovery of a novel member of the genus phlebovirus. J Virol. 2015 Jan;89(1):594-604. DOI 10.1128/JVI.02704-14.
(10.) Floris R, Altobelli A, Boemo B, Mignozzi K, Cinco M. First detection of TBE virus sequences in Ixodes ricinus from Friuli Venezia Giulia (Italy). New Microbiol. 2006 Apr;29(2):147-50.
(11.) Han X, Aho M, Vene S, Peltomaa M, Vaheri A, Vapalahti O. Prevalence of tick-borne encephalitis virus in Ixodes ricinus ticks in Finland. J Med Virol. 2001 May;64(1):21-8.
(12.) Katargina O, Russakova S, Geller J, Kondrusik M, Zajkowska J, Zygutiene M, et al. Detection and characterization of tick-borne encephalitis virus in Baltic countries and eastern Poland. PLoS One. 2013 May;8(5):e61374. DOI 10.1371/journal.pone.0061374.
(13.) Klaus C, Hoffmann B, Hering U, Mielke B, Sachse K, Beer M, et al. Tick-borne encephalitis (TBE) virus prevalence and virus genome characterization in field-collected ticks (Ixodes ricinus) from risk, non-risk and former risk areas of TBE, and in ticks removed from humans in Germany. Clin Microbiol Infect. 2010 Mar;16(3):238-44. DOI 10.1111/j.1469-0691.2009.02764.x.
(14.) Saksida A, Duh D, Lotric-Furlan S, Strle F, Petrovec M, Avsic-Zupanc T. The importance of tick-borne encephalitis virus RNA detection for early differential diagnosis of tick-borne encephalitis. J Clin Virol. 2005 Aug;33(4):331-5. DOI 10.1016/j.jcv.2004.07.014.
(15.) Fakoorziba MR, Golmohammadi P, Moradzadeh R, Moemenbellah-Fard MD, Azizi K, Davari B, et al. Reverse transcription PCR-based detection of Crimean-Congo hemorrhagic fever virus isolated from ticks of domestic ruminants in Kurdistan province of Iran. Vector Borne Zoonotic Dis. 2012 Sep;12(9):794-9. DOI 10.1089/vbz.2011.0743.
(16.) Mehravaran A, Moradi M, Telmadarraiy Z, Mostafavi E, Moradi AR, Khakifirouz S, et al. Molecular detection of Crimean-Congo haemorrhagic fever (CCHF) virus in ticks from southeastern Iran. Ticks Tick Borne Dis. 2013 Feb;4(1-2):35-8. DOI 10.1016/j.ttbdis.2012.06.006.
(17.) Lindquist L, Vapalahti O. Tick-borne encephalitis. Lancet. 2008 May;371(9627):1861-71. DOI 10.1016/S0140-6736(08)60800-4.
(18.) Bröker M, Kollaritsch H. After a tick bite in a tickborne encephalitis virus endemic area: current positions about post-exposure treatment. Vaccine. 2008 Feb;26(7):863-8. DOI 10.1016/j.vaccine.2007.11.046.
(19.) Bogovic P, Strle F. Tick-borne encephalitis: A review of epidemiology, clinical characteristics, and management. World J Clin Cases. 2015 May;3(5):430-41. DOI 10.12998/wjcc.v3.i5.430.
(20.) Donoso Mantke O, Schädler R, Niedrig M. A survey on cases of tick-borne encephalitis in European countries. Euro Surveill. 2008 Apr;13(17). pii: 18848.
(21.) Günther G, Lindquist L. Surveillance of tick-borne encephalitis in Europe and case definition. Euro Surveill. 2005 Jan;10(1):2-3.
(22.) Stefanoff P, Eidson M, Morse DL, Zielinski A. Evaluation of tickborne encephalitis case classification in Poland. Euro Surveill. 2005 Jan;10(1):23-5.
(23.) Hubálek Z, Rudolf I. Tick-borne viruses in Europe. Parasitol Res. 2012 Jul;111(1):9-36. DOI 10.1007/s00436-012-2910-1.
(24.) Charrel RN, Attoui H, Butenko AM, Clegg JC, Deubel V, Frolova TV, et al. Tick-borne virus diseases of human interest in Europe. Clin Microbiol Infect. 2004 Dec;10(12):1040-55. DOI 10.1111/j.1469-0691.2004.01022.x.
(25.) Dumpis U, Crook D, Oksi J. Tick-borne encephalitis. Clin Infect Dis. 1999 Apr;28(4):882-90.
(26.) Lani R, Moghaddam E, Haghani A, Chang LY, Abu-Bakar S, Zandi K. Tick-borne viruses: a review from the perspective of therapeutic approaches. Ticks Tick Borne Dis. 2014 Sep;5(5):457-65. DOI 10.1016/j.ttbdis.2014.04.001.
(27.) Loew-Baselli A, Poellabauer EM, Pavlova BG, Fritsch S, Firth C, Petermann R, et al. Prevention of tick-borne encephalitis by FSME-IMMUN vaccines: review of a clinical development programme. Vaccine. 2011 Oct;29(43):7307-19. DOI 10.1016/j.vaccine.2011.07.089.
(28.) Lehrer AT, Holbrook MR. Tick-borne Encephalitis Vaccines. J Bioterror Biodef. 2011;2011(Suppl 1):3. DOI 10.4172/2157-2526.S1-003.
(29.) Calisher CH. Antigenic classification and taxonomy of flaviviruses (family Flaviviridae) emphasizing a universal system for the taxonomy of viruses causing tick-borne encephalitis. Acta Virol. 1988 Sep;32(5):469-78.
(30.) Gritsun TS, Nuttall PA, Gould EA. Tick-borne flaviviruses. Adv Virus Res. 2003;61:317-71. DOI 10.1016/j.cll.2010.01.002.
(31.) Centers for Disease Control and Prevention (CDC). Morbidity and Mortality Weekly Report (MMWR): West Nile virus disease and other arboviral diseases – United States, 2011. Morbidity and Mortality Weekly Report [Internet] 2012 [cited 2016 Nov 25];61(27):[510-4]. Available from: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6127a3.htm
(32.) McLean D, Donahue W. Powassan virus: isolation of virus from a fatal case of encephalitis. Can Med Assoc J. 1959 May;80(9):708-11.
(33.) Thomas L, Kennedy R, Eklund C. Isolation of a virus closely related to Powassan virus from Dermacentor andersoni collected along North Cache la Poudre River, Colo. Proc Soc Exp Biol Med. 1960 Jun;104:355-9.
(34.) Eldridge B, Scott T, Day J, Tabachnick W. Arbovirus Diseases. In: Eldridge B, Edman J, editores. Medical Entomology, 2nd ed. Davis (CA): Springer; 2004. p. 415-60.
(35.) Růžek D, Yakimenko VV, Karan LS, Tkachev SE. Omsk haemorrhagic fever. Lancet. 2010 Dec;376(9758):2104-13. DOI 10.1016/S0140-6736(10)61120-8.
(36.) Centers for Disease Control and Prevention [Internet]. Atlanta: CDC; 2013 [cited 2016 Nov 25]. Omsk Hemorrhagic Fever (OHF): Treatment. Available from: http://www.cdc.gov/vhf/omsk/treatment/index.html
(37.) Tandale BV, Balakrishnan A, Yadav PD, Marja N, Mourya DT. New focus of Kyasanur Forest disease virus activity in a tribal area in Kerala, India, 2014. Infect Dis Poverty. 2015 Mar;4:12. DOI 10.1186/s40249-015-0044-2.
(38.) Holbrook MR. Kyasanur forest disease. Antiviral Res. 2012 Dec;96(3):353-62. DOI 10.1016/j.antiviral.2012.10.005.
(39.) Dodd KA, Bird BH, Khristova ML, Albariño CG, Carroll SA, Comer JA, et al. Ancient ancestry of KFDV and AHFV revealed by complete genome analyses of viruses isolated from ticks and mammalian hosts. PLoS Negl Trop Dis. 2011 Oct;5(10):e1352. DOI 10.1371/journal.pntd.0001352.
(40.) Hoogstraal H. The epidemiology of tick-borne Crimean-Congo hemorrhagic fever in Asia, Europe, and Africa. J Med Entomol. 1979 May;15(4):307-417.
(41.) Madder M, Pascucci I. Factors influencing the spread and distribution of ticks. In: Salman M, Tarrés-Call J, editores. Ticks and tick-borne diseases: Geographical distribution and control strategies in the Euro-Asia region. United Kingdom: CAB International; 2013. p. 27-32.
(42.) Ergönül O. Crimean-Congo haemorrhagic fever. Lancet Infect Dis. 2006 Apr;6(4):203-14. DOI 10.1016/S1473-3099(06)70435-2.
(43.) Salata C, Calistri A, Parolin C, Palù G. Summary of the 9th annual meeting of the Italian Society for Virology. J Cell Physiol. 2011 Jan;226(1):285-7. DOI 10.1002/jcp.22396.
(44.) Celik VK, Sari I, Engin A, Gürsel Y, Aydin H, Bakir S. Determination of serum adenosine deaminase and xanthine oxidase levels in patients with crimean-congo hemorrhagic fever. Clinics (Sao Paulo). 2010 Jul;65(7):697-702. DOI 10.1590/S1807-59322010000700008.
(45.) Keshtkar-Jahromi M, Kuhn JH, Christova I, Bradfute SB, Jahrling PB, Bavari S. Crimean-Congo hemorrhagic fever: current and future prospects of vaccines and therapies. Antiviral Res. 2011 May;90(2):85-92. DOI 10.1016/j.antiviral.2011.02.010.
(46.) Hubálek Z. Biogeography of tick-borne bhanja virus (bunyaviridae) in europe. Interdiscip Perspect Infect Dis. 2009;2009:372691. DOI 10.1155/2009/372691.
(47.) Elliott RM, Brennan B. Emerging phleboviruses. Curr Opin Virol. 2014 Apr;5:50-7. DOI 10.1016/j.coviro.2014.01.011.
(48.) Matsuno K, Weisend C, Travassos da Rosa AP, Anzick SL, Dahlstrom E, Porcella SF, et al. Characterization of the Bhanja serogroup viruses (Bunyaviridae): a novel species of the genus Phlebovirus and its relationship with other emerging tick-borne phleboviruses. J Virol. 2013 Apr;87(7):3719-28. DOI 10.1128/JVI.02845-12.
(49.) Dilcher M, Alves MJ, Finkeisen D, Hufert F, Weidmann M. Genetic characterization of Bhanja virus and Palma virus, two tick-borne phleboviruses. Virus Genes. 2012 Oct;45(2):311-5. DOI 10.1007/s11262-012-0785-y.
(50.) Li DX. Severe fever with thrombocytopenia syndrome: a newly discovered emerging infectious disease. Clin Microbiol Infect. 2015 Jul;21(7):614-20. DOI 10.1016/j.cmi.2015.03.001.
(51.) Liu Q, He B, Huang SY, Wei F, Zhu XQ. Severe fever with thrombocytopenia syndrome, an emerging tickborne zoonosis. Lancet Infect Dis. 2014 Aug;14(8):763-72. DOI 10.1016/S1473-3099(14)70718-2.
(52.) Liu K, Zhou H, Sun RX, Yao HW, Li Y, Wang LP, et al. A national assessment of the epidemiology of severe fever with thrombocytopenia syndrome, China. Sci Rep. 2015 Apr;5:9679. DOI 10.1038/srep09679.
(53.) Palmarini M. Reoviridae. In: MacLachlan J, Dubovi E, editores. Fenner’s Veterinary Virology. 5th ed. Chennai: Academic Press; 2016. p. 299-317.
(54.) Ejiri H, Lim CK, Isawa H, Kuwata R, Kobayashi D, Yamaguchi Y, et al. Genetic and biological characterization of Muko virus, a new distinct member of the species Great Island virus (genus Orbivirus, family Reoviridae), isolated from ixodid ticks in Japan. Arch Virol. 2015 Dec;160(12):2965-77. DOI 10.1007/s00705-015-2588-7.
(55.) Attoui H, Mohd Jaafar F, Biagini P, Cantaloube JF, de Micco P, Murphy FA, et al. Genus Coltivirus (family Reoviridae): genomic and morphologic characterization of Old World and New World viruses. Arch Virol. 2002 Mar;147(3):533-61. DOI 10.1007/s007050200005.
(56.) Libíková H, Heinz F, Ujházyová D, Stünzner D. Orbiviruses of the Kemerovo complex and neurological diseases. Med Microbiol Immunol. 1978 Nov;166(1-4):255-63. DOI 10.1007/BF02121159.
(57.) Yendell SJ, Fischer M, Staples JE. Colorado tick fever in the United States, 2002-2012. Vector Borne Zoonotic Dis. 2015 May;15(5):311-6. DOI 10.1089/vbz.2014.1755.
(58.) Geissler AL, Thorp E, Van Houten C, Lanciotti RS, Panella N, Cadwell BL, et al. Infection with Colorado tick fever virus among humans and ticks in a national park and forest, Wyoming, 2010. Vector Borne Zoonotic Dis. 2014 Sep;14(9):675-80. DOI 10.1089/vbz.2013.1568.
(59.) Mohd Jaafar F, Attoui H, De Micco P, De Lamballerie X. Recombinant VP6-based enzyme-linked immunosorbent assay for detection of immunoglobulin G antibodies to Eyach virus (genus Coltivirus). J Clin Virol. 2004 Jul;30(3):248-53. DOI 10.1016/j.jcv.2003.12.004.
(60.) Keita D, Heath L, Albina E. Control of African swine fever virus replication by small interfering RNA targeting the A151R and VP72 genes. Antivir Ther. 2010;15(5):727-36. DOI 10.3851/IMP1593.
(61.) Costard S, Mur L, Lubroth J, Sanchez-Vizcaino JM, Pfeiffer DU. Epidemiology of African swine fever virus. Virus Res. 2013 Apr;173(1):191-7. DOI 10.1016/j.virusres.2012.10.030.
(62.) Briese T, Chowdhary R, Travassos da Rosa A, Hutchison SK, Popov V, Street C, et al. Upolu virus and Aransas Bay virus, two presumptive bunyaviruses, are novel members of the family Orthomyxoviridae. J Virol. 2014 May;88(10):5298-309. DOI 10.1128/JVI.03391-13.
(63.) Kosoy OI, Lambert AJ, Hawkinson DJ, Pastula DM, Goldsmith CS, Hunt DC, et al. Novel thogotovirus associated with febrile illness and death, United States, 2014. Emerg Infect Dis. 2015 May;21(5):760-4. DOI 10.3201/eid2105.150150.
(64.) Bussetti AV, Palacios G, Travassos da Rosa A, Savji N, Jain K, Guzman H, et al. Genomic and antigenic characterization of Jos virus. J Gen Virol. 2012 Feb;93(Pt 2):293-8. DOI 10.1099/vir.0.035121-0.
(65.) Guilligay D, Kadlec J, Crépin T, Lunardi T, Bouvier D, Kochs G, et al. Comparative structural and functional analysis of orthomyxovirus polymerase cap-snatching domains. PLoS One. 2014 Jan;9(1):e84973. DOI 10.1371/journal.pone.0084973.
(66.) Contreras-Gutiérrez MA, Nunes MR, Guzman H, Uribe S, Gallego Gómez JC, Suaza Vasco JD, et al. Sinu virus, a novel and divergent orthomyxovirus related to members of the genus Thogotovirus isolated from mosquitoes in Colombia. Virology. 2017 Jan;501:166-175. DOI 10.1016/j.virol.2016.11.014.
(67.) Guglielmone AA, Apanaskevich DA, Estrada-Peña A, Robbins RG, Petney TN, Horak IG. The hard ticks of the world: (Acari: Ixodida: Ixodidae). New York: Springer; 2014.
(68.) de Figueiredo GG, Amarilla AA, de Souza WM, Fumagalli MJ, de Figueiredo MLG, Szabó MPJ, et al. Genetic characterization of Cacipacoré virus from ticks collected in São Paulo State, Brazil. Arch Virol. 2017 Jun;162(6):1783-6. DOI 10.1007/s00705-017-3279-3.
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2018 Universidad de Antioquia

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-CompartirIgual 4.0.
Los artículos publicados en la revista están disponibles para ser utilizados bajo la licencia Creative Commons, específicamente son de Reconocimiento-NoComercial-CompartirIgual 4.0 Internacional.
Los trabajos enviados deben ser inéditos y suministrados exclusivamente a la Revista; se exige al autor que envía sus contribuciones presentar los formatos: presentación de artículo y responsabilidad de autoría completamente diligenciados.