Intravitreal ranibizumab injections in patients with retinopathy of prematurity (ROP)
DOI:
https://doi.org/10.17533/udea.iatreia.v31n1a03Keywords:
antiangiogenics, intravitreal injections, retinopathy of prematurityAbstract
Purpose: To describe ocular outcomes after intravitreal ranibizumab injections in patients with retinopathy of prematurity (ROP), who had transpupillary laser thermotherapy without regression of the disease.
Methods: Historical cohort in Clínica Universitaria Bolivariana (CUB) during 2008-2015. Evaluation of patients with aggressive posterior ROP, threshold disease, who received intravitreal ranibizumab secondary to treatment with transpupillary thermotherapy. The primary outcome was treatment success defined as the resolution of neovascularization (NV) and no recurrence. The secondary endpoints were visual acuity and ocular and systemic complications.
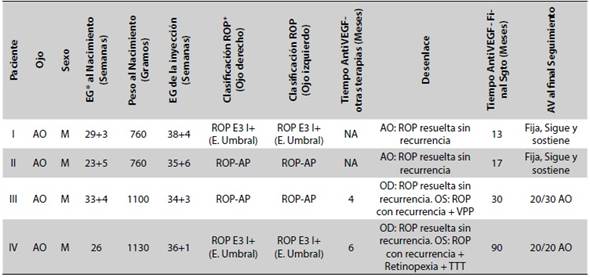
Results: Eight eyes of four patients were included in the study. Six eyes had resolution of retinopathy, Incidence density 48 x 100 person years, 95 % CI = 17.6 - 100 with a median follow-up of 23.5 months, range 13-90 months. No ocular or systemic complications associated with the procedure was found.
Conclusion: This cohort suggests that intravitreal injection of ranibizumab for ROP in refractory laser photocoagulation cases, results in apparent ocular preservation a long-term the outcome.
Downloads
References
(1.) Clark D, Mandal K. Treatment of retinopathy of prematurity. Early Hum Dev. 2008 Feb;84(2):95-9. DOI 10.1016/j.earlhumdev.2007.11.007.
(2.) Gergely K, Gerinec A. Retinopathy of prematurity--epidemics, incidence, prevalence, blindness. Bratisl Lek Listy. 2010;111(9):514-7.
(3.) Giraldo M, Hurtado A, Donado J, Molina M. Epidemiología de la retinopatía del prematuro en Medellín, 2003-2008. Iatreia. 2011;24 Jul-Sept;(3):250-8.
(4.) Zin A. The increasing problem of retinopathy of prematurity. J Comm Eye Health 2001; 14(40): 58-59.
(5.) Castellanos MA, Schwartz S, García-Aguirre G, Quiroz-Mercado H. Short-term outcome after intravitreal ranibizumab injections for the treatment of retinopathy of prematurity. Br J Ophthalmol. 2013 Jul;97(7):816-9. DOI 10.1136/bjophthalmol-2012-302276.
(6.) BOOST II United Kingdom Collaborative Group; BOOST II Australia Collaborative Group; BOOST II New Zealand Collaborative Group, Stenson BJ, Tarnow-Mordi WO, Darlow BA, et al. Oxygen saturation and outcomes in preterm infants. N Engl J Med. 2013 May;368(22):2094-104. DOI 10.1056/NEJMoa1302298.
(7.) Hartnett ME, Penn JS. Mechanisms and management of retinopathy of prematurity. N Engl J Med. 2012 Dec;367(26):2515-26. DOI 10.1056/NEJMra1208129.
(8.) O’Keefe M, Kirwan C. Screening for retinopathy of prematurity. Early Hum Dev. 2008 Feb;84(2):89-94. DOI 10.1016/j.earlhumdev.2007.11.006.
(9.) Eltzschig HK, Carmeliet P. Hypoxia and inflammation. N Engl J Med. 2011 Feb ;364(7):656-65. DOI 10.1056/NEJMra0910283.
(10.) Good WV; Early Treatment for Retinopathy of Prematurity Cooperative Group. Final results of the Early Treatment for Retinopathy of Prematurity (ETROP) randomized trial. Trans Am Ophthalmol Soc. 2004;102:233-48; discussion 248-50.
(11.) International Committee for the Classification of Retinopathy of Prematurity. The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol. 2005 Jul;123(7):991-9.
(12.) Centro Nacional de Investigación en Evidencia y Tecnologías en Salud CINETS. Guía de práctica clínica del recién nacido prematuro - 2013 Guía No. 04 [Internet]. Bogotá: Ministerio de Salud y Protección Social – Colciencias; 2013. Disponible en: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/INEC/IETS/GPC_Completa_Premat.pdf
(13.) Mintz-Hittner HA, Kennedy KA, Chuang AZ; BEAT-ROP Cooperative Group. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Engl J Med. 2011 Feb;364(7):603-15. DOI 10.1056/NEJMoa1007374.
(14.) Menke MN, Framme C, Nelle M, Berger MR, Sturm V, Wolf S. Intravitreal ranibizumab monotherapy to treat retinopathy of prematurity zone II, stage 3 with plus disease. BMC Ophthalmol. 2015 Mar;15:20. DOI 10.1186/s12886-015-0001-7.
(15.) Jabs DA. Improving the reporting of clinical case series. Am J Ophthalmol. 2005 May;139(5):900-5.
(16.) Arámbulo O, Dib G, Iturralde J, Duran F, Brito M, Fortes Filho JB. Intravitreal ranibizumab as a primary or a combined treatment for severe retinopathy of prematurity. Clin Ophthalmol. 2015 Oct;9:2027-32. DOI 10.2147/OPTH.S90979.
(17.) Erol MK, Coban DT, Sari ES, Bilgin AB, Dogan B, Ozdemir O, et al. Comparison of intravitreal ranibizumab and bevacizumab treatment for retinopathy of prematurity. Arq Bras Oftalmol. 2015 Nov-Dec;78(6):340-3. DOI 10.5935/0004-2749.20150090.
(18.) Yi Z, Su Y, Zhou Y, Zheng H, Ye M, Xu Y, et al. Effects of Intravitreal Ranibizumab in the Treatment of Retinopathy of Prematurity in Chinese Infants. Curr Eye Res. 2016 Aug;41(8):1092-1097.
(19.) Menke MN, Framme C, Nelle M, Berger MR, Sturm V, Wolf S. Intravitreal ranibizumab monotherapy to treat retinopathy of prematurity zone II, stage 3 with plus disease. BMC Ophthalmol. 2015 Mar;15:20. DOI 10.1186/s12886-015-0001-7.
(20.) Mintz-Hittner HA, Geloneck MM. Review of effects of anti-VEGF treatment on refractive error. Eye Brain. 2016 Jun;8:135-140. DOI 10.2147/EB.S99306.
(21.) Jang SY, Choi KS, Lee SJ. Delayed-onset retinal detachment after an intravitreal injection of ranibizumab for zone 1 plus retinopathy of prematurity. J AAPOS. 2010 Oct;14(5):457-9. DOI 10.1016/j.jaapos.2010.05.011.
(22.) Honda S, Hirabayashi H, Tsukahara Y, Negi A. Acute contraction of the proliferative membrane after an intravitreal injection of bevacizumab for advanced retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol. 2008 Jul;246(7):1061-3. DOI 10.1007/s00417-008-0786-7.
(23.) Mota A, Carneiro A, Breda J, Rosas V, Magalhães A, Silva R, et al. Combination of intravitreal ranibizumab and laser photocoagulation for aggressive posterior retinopathy of prematurity. Case Rep Ophthalmol. 2012 Jan;3(1):136-41. DOI 10.1159/000338623.
(24.) Baumal CR, Goldberg RA, Fein JG. Primary intravitreal ranibizumab for high-risk retinopathy of prematurity. Ophthalmic Surg Lasers Imaging Retina. 2015 Apr;46(4):432-8. DOI 10.3928/23258160-20150422-05.
Published
How to Cite
Issue
Section
License
Copyright (c) 2018 Universidad de Antioquia

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Papers published in the journal are available for use under the Creative Commons license, specifically Attribution-NonCommercial-ShareAlike 4.0 International.
The papers must be unpublished and sent exclusively to the Journal Iatreia; the author uploading the contribution is required to submit two fully completed formats: article submission and authorship responsibility.